In recent years, the growing demand for antioxidant and cell-protective ingredients has brought ergothioneine (EGT) into the spotlight across the dietary supplement, functional food, and cosmetic industries.
Despite its increasing popularity, achieving high-efficiency and cost-effective large-scale production of ergothioneine remains a major challenge for manufacturers. Limitations in natural biosynthetic pathways, complex precursor metabolism, and fermentation costs have all constrained its broader industrial application.
A recent study published in the Journal of Agricultural and Food Chemistry explored new strategies to improve EGT yields through metabolic engineering and fermentation optimization. The findings provide valuable insights and a promising technical route toward more sustainable and scalable industrial production.
Ergothioneine is a naturally occurring sulfur-containing derivative of the amino acid histidine. It has attracted growing interest in recent years due to several unique properties:
strong antioxidant activity
excellent stability under heat and a wide pH range
selective uptake in the human body via the OCTN1 transporter
Because the human body cannot synthesize ergothioneine on its own, it must be obtained through dietary sources or supplementation. A growing body of research suggests that EGT may have potential applications in:
antioxidant and anti-inflammatory formulations
neuroprotection and healthy aging products
skin repair and anti-photoaging cosmetics
Following its approval as a novel food ingredient in the European Union, the global market for ergothioneine is expected to expand steadily in the coming years. This regulatory progress has also accelerated demand for high-purity ergothioneine powder suitable for large-scale commercial use.
To improve microbial production of EGT, researchers isolated key biosynthetic genes from Methylobacterium aquaticum and reconstructed the pathway in Escherichia coli. The engineered pathway relies on three core enzymes:
EgtB
EgtD
EgtE
By adjusting plasmid copy numbers to fine-tune enzyme expression levels, the research team was able to enhance product accumulation. As a result, EGT titers increased from 29.5 mg/L to 35 mg/L, providing a stronger foundation for further metabolic engineering efforts.
These findings highlight the importance of balancing metabolic flux through precise control of gene expression, which remains a critical strategy for improving ergothioneine fermentation yields.
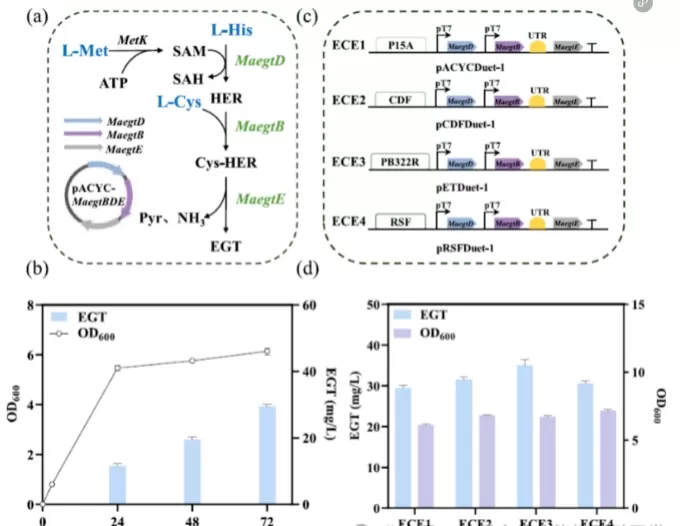
Figure 1. Design and optimization of the EGT biosynthetic pathway.(a) Reconstructed ergothioneine biosynthetic pathway derived from Methylobacterium aquaticum DSM 16371.(b) Time-course profiles of EGT production and cell growth (OD600) in strain ECE1.(c) Schematic illustration of plasmid construction used for copy number optimization.(d) Comparison of EGT titers and OD600 values among strains harboring plasmids with different copy numbers.
The biosynthesis of ergothioneine relies on three key amino acid precursors:
histidine
cysteine
methionine
To further enhance EGT yields, the research team applied targeted metabolic engineering strategies to increase intracellular precursor availability. This was achieved by removing feedback inhibition and redirecting metabolic flux toward amino acid biosynthesis.
Key modifications included:
deletion of metJ, a transcriptional repressor, to enhance methionine biosynthesis
introduction of feedback-resistant variants metA^mut and thrA to improve amino acid flux
deletion of sdaA to reduce serine degradation and support cysteine formation
Following these combined genetic modifications, EGT titers increased to 130 mg/L, representing a several-fold improvement over the original strain.
From an industrial biotechnology perspective, these results underscore that optimizing precursor supply is one of the most effective strategies for improving the efficiency of microbial ergothioneine production.
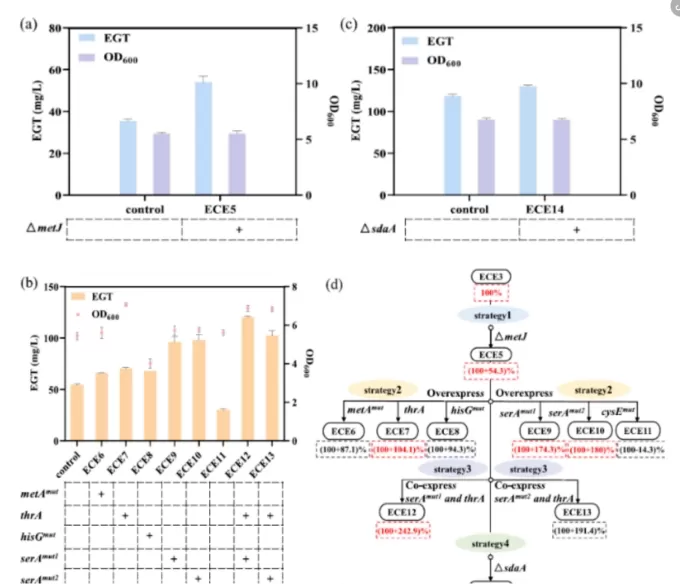
Figure 2. Metabolic engineering of precursor amino acids to enhance EGT production.(a) Effect of metJdeletion on EGT titers.(b) Impact of gene overexpression and co-expression on EGT production.(c) Effect of sdaA deletion on EGT accumulation.(d) Influence of increased precursor amino acid supply on EGT yields.
Building on the optimized strains, the research team further refined the fermentation process:
Identified 20 g/L as the optimal glucose concentration
Replaced yeast extract with corn steep liquor (CSL) to reduce production costs
Using fed-batch fermentation in a 10 L bioreactor, the process achieved:
EGT titer: 595 mg/L
Productivity: 8.2 mg/L/h
These results demonstrate a significant balance between cost efficiency and production yield, providing a practical route for commercial-scale ergothioneine manufacturing.
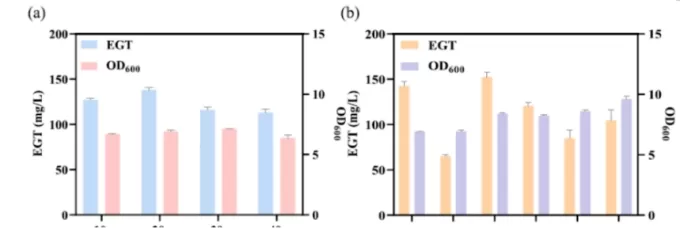
Figure 3: Optimization of the fermentation process for strain ECE14. (a) EGT production at different glucose concentrations. (b) Evaluation of using corn steep liquor (CSL) to replace yeast extract (YE) at various concentrations.
This research delivers three major signals for the ergothioneine market:
1. EGT fermentation technology is maturing
The integration of metabolic engineering with process optimization makes microbial fermentation the most competitive route for ergothioneine powder production.
2. Raw material costs can be further reduced
Using industrial by-products like corn steep liquor (CSL) instead of yeast extract lowers the cost of bulk ergothioneine, making large-scale production more economically feasible.
3. Stable supply of high-purity EGT
As fermentation pathways improve, the industry can gradually move away from natural extraction, ensuring a consistent and scalable supply of EGT.
For dietary supplement brands, functional food companies, and cosmetic formulators, this trend highlights that ergothioneine powder is increasingly a commercially viable functional ingredient.
With growing global demand for antioxidants and cell-protective ingredients, brands prioritize partners who can provide reliable, high-purity ergothioneine.
Natural Field specializes in functional active ingredients and offers:
Premium ergothioneine powder with high purity
Consistent quality across all production batches
Raw materials suitable for dietary supplements and cosmetic formulations
As an experienced ergothioneine supplier and EGT manufacturer, we focus not only on scale but also on:
Fermentation process optimization
Raw material traceability
Customizable purity and specifications
If you are looking for a trusted partner for bulk ergothioneine or want to explore EGT applications in your product line, our technical team can provide detailed information and samples to support your development.